Unmet Clinical Need
Tramadol is a popular pain medication prescribed over 40 million times each year in the United States for moderate to severe acute, chronic and neuropathic pain. Tramadol is believed to be safer and less addictive than schedule II opioids such as oxycodone and tapentadol [1], and its use has increased significantly. Tragically, millions of patients do not obtain pain relief from tramadol because they are unable to metabolize the drug to its active form. In some patient populations, 1 in 3 patients will not obtain adequate pain relief from tramadol either because they are also taking one of many common antidepressants that compete for the enzyme that metabolizes tramadol to its active form, or because they have a genetic variant in the enzyme. [2]. The need to switch non-responders to schedule II opioids increases the risk of developing addiction and adds costs and inconvenience. Still other patients metabolize tramadol too quickly, causing rare but life-threatening side effects that include respiratory depression. There is therefore a critical need for a new tramadol-like pain medication that is effective without requiring metabolism, but that enjoys the same safety and lower addictive potential as tramadol.
Omnitram is Developed
To meet this critical need, Syntrix developed Omnitram, a proprietary solution to tramadol failure that provides the body with the primary active metabolite of tramadol, O-desmethyltramadol, in a precise timed delivery without requiring metabolic activation. Pain relief using Omnitram results from a dual mechanism involving mu-opioid receptor activation and inhibition of the reuptake of norepinephrine (see below).
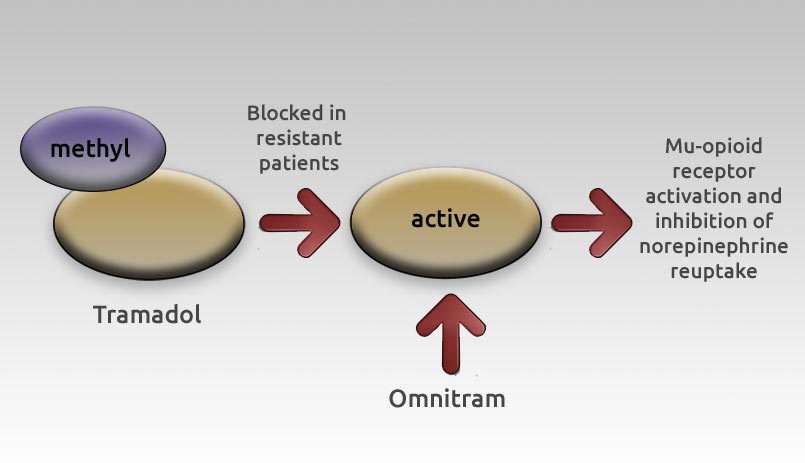
When Syntrix initially began its program to create a new and improved tramadol-like pain medication, it unexpectedly discovered that it was impossible to simply administer the active metabolite of tramadol directly; doing so resulted in rapid clearance of the molecule and wide variation in blood levels. Overcoming this setback required Syntrix go back to the drawing board. For over a year company scientists tirelessly dedicated themselves to finding a way to correctly deliver the active metabolite to resistant patients. After countless formulations and thousands of samples tested, Omnitram was finally born.
Omnitram’s MetabX™
The technology replicates ideal liver metabolism by controlling the rates of hydration, dissolution, and erosion in the stomach.
Result:
Omnitram delivers the M1 metabolite directly and consistently in a manner that does not require metabolic activation by CYP2D6.
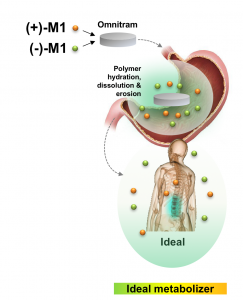
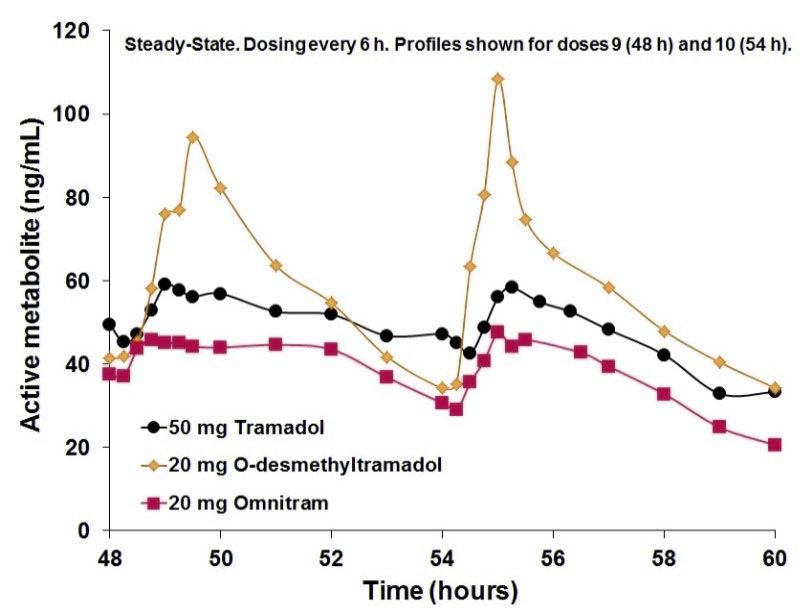
Omnitram is a novel innovative formula that succeeds in releasing the active metabolite in a precise temporal formula that tricks the drug into adopting a blood profile that matches the active metabolite profile from tramadol (see below).
Omnitram is remarkable in that it replicates blood levels of the active tramadol metabolite but without requiring metabolism, thus providing pain relief in patients who would otherwise fail to obtain pain relief from tramadol, as well as preventing dangerously high blood levels of the active metabolite in patients whose genetic make-up causes them to metabolize tramadol excessively fast.
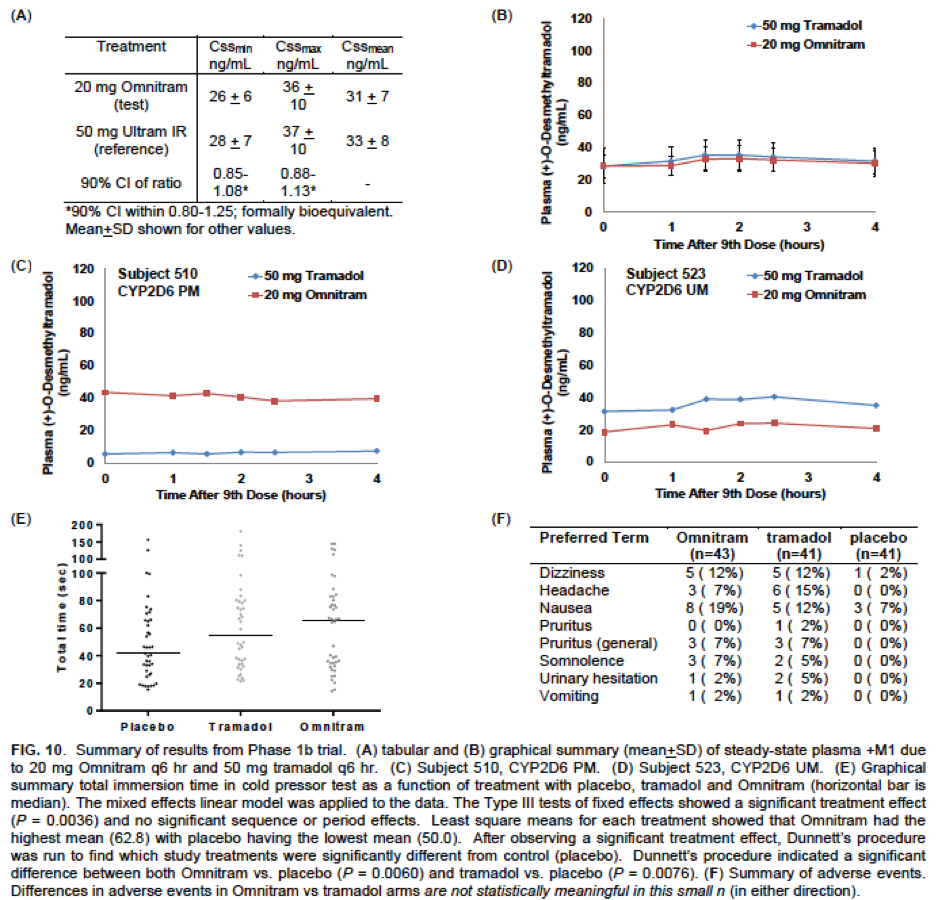
Successful double-blind randomized Phase 1B trial: Safe and effective with analgesia as good as tramadol
43 patient, double-blind, randomized controlled double crossover study evidence of:
- Matching active M1 PK profile to tramadol (A, B)
Omnitram rescued metabolic defects in:
- Poor Metabolizer (C)
- Ultra Metabolizer (D)
- Effective analgesia (P<.01) (E)
- Safe (F)
The Omnitram Market
The global market for Omnitram is over $2 billion. Omnitram targets the existing world opioid and tramadol markets with a “better” tramadol. Retail U.S. sales of just the generic forms of hydrocodone, oxycodone, fentanyl and tramadol total over $6.1 billion. Tramadol itself is widely prescribed, with over 40 million prescriptions in the U.S. alone. Globally, tramadol sales are well over $2.0 billion. Omnitram’s dual mechanism of pain relief is expected to be effective in treating neuropathic pain, a type of pain that is particularly difficult to treat with current therapies. Opioids are frequently ineffective and other treatments such as gabapentin (Neurontin™) are associated with significant side effects. No current single drug treatment is effective in more than half of patients, and therefore novel therapeutic approaches are urgently needed. Approximately 26 million patients are affected worldwide and the global market for neuropathic pain is projected to reach $18 billion by 2020.
[1] Tsutaoka BT, Ho RY, Fung SM and Kearney TE. Comparative toxicity of tapentadol and tramadol utilizing data reported to the National Poison Data System [of the American Association of Poison Control Centers]. Annals of Pharmacotherapy 2015 Sep 14; pii: 1060028015604631. [Epub ahead of print]
[2] Preskorn SH, Kane CP, Lobello K, Nichols AI, Fayyad R, Buckley G, Focht K, and Guico-Pabia CJ. Cytochrome P450 2D6 phenoconversion is common in patients being treated for depression: Implications for personalized medicine. J Clin Psychiatry 2013 Jun; 74(6):614-21
Unmet Clinical Need
It has recently become clear that the presence of myeloid-derived suppressor cells (MDSCs) in human tumors has the most adverse prognostic significance of any infiltrating cell type. MDSCs serve the tumor as a “cloak” from the patient’s immune system, assisting in metastasis, and mediating resistance to chemotherapy and anti-PD1 therapy.
SX-682 is a potent oral clinical-stage small-molecule antagonist of the chemokine receptors CXCR1 and CXCR2 (“CXCR1/2”), the receptors pivotal to the recruitment of MDSCs by tumors. Blocking the CXCR1/2 pathway operates “upstream” of all the other biology in those cells by simply keep the immunosuppressive MDSCs out of the tumor microenvironment, enabling a t-cell response.
In collaborations with a host of top academic collaborators SX-682 has demonstrated activity mouse tumor models in two models of breast cancer, two models of melanoma, lung cancer, and most recently in prostate cancer, where it potently activated PD-1 and CTL4 in a tumor otherwise unresponsive to immune checkpoint inhibition, work recently published in Nature. ( doi: 10.1038./nature21676 )
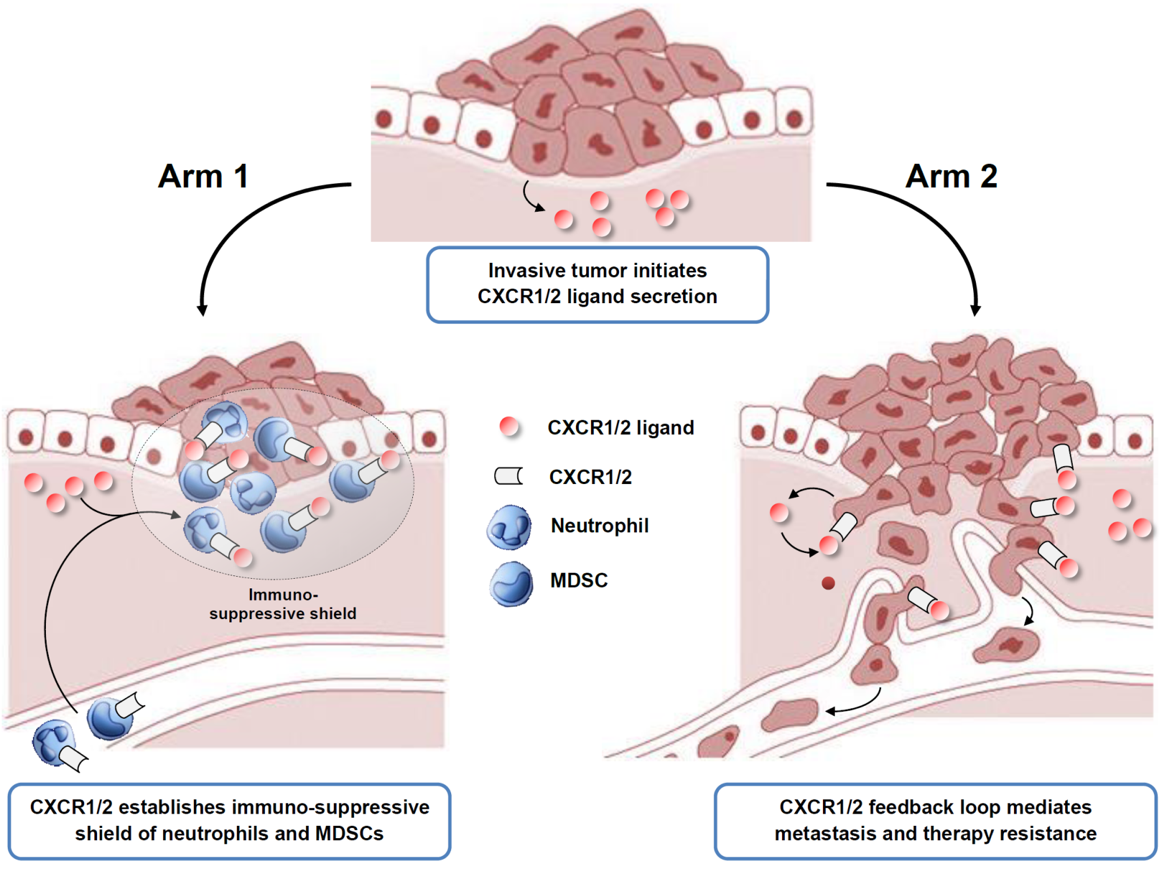
Best-In-Class Immuno-Oncology (I/O) Therapy for Abrogating the Tumor Immunosuppressive Microenvironment and Blocking Metastasis
Tumors secrete chemo-attractive proteins that act via chemokine receptors CXCR1 and CXCR2 (“CXCR1/2”) to recruit immunosuppressive neutrophils (“PMNs”) and myeloid-derived-suppressor-cells (“MDSCs”) to the tumor microenvironment, hiding the tumor from cytolytic effector T cells and natural killer cells (“NK cells”) of the adaptive immune system. Tumor-secreted chemokines also act directly on the tumor cell’s own CXCR1/2 receptors to drive of metastasis and resistance to chemotherapeutic and immunologic therapies. CXCR1/2 is validated I/O target with major programs in the clinic at Bristol-Meyer Squibb and AstraZeneca.
Syntrix’s SX-682 is a novel oral small-molecule dual-inhibitor of both CXCR1 and CXCR2. By inhibiting CXCR1/2 it abrogates pro-tumorigenic PMN/MDSC microenvironment, unmasking the tumor to killing by the adaptive immune system and blocking metastasis. As a dual-inhibitor of both receptors SX-682 is best-in-class. SX-682 has been validated in numerous tumor models, where it exhibits single agent activity, synergizes with checkpoint inhibitors and chemotherapy,and blocks metastasis. SX-682 has completed GLP toxicology, demonstrating an exceedingly safe profile with no dose-limiting toxicities. SX-682 has an open IND with FDA for a Phase I/II trial in melanoma in combination with anti-PD-1 at Mass General Hospital (clinicaltrials.gov: NCT03161431) and NIH funding to conduct this trial. SX-682 is covered by patents out to at least 2033.
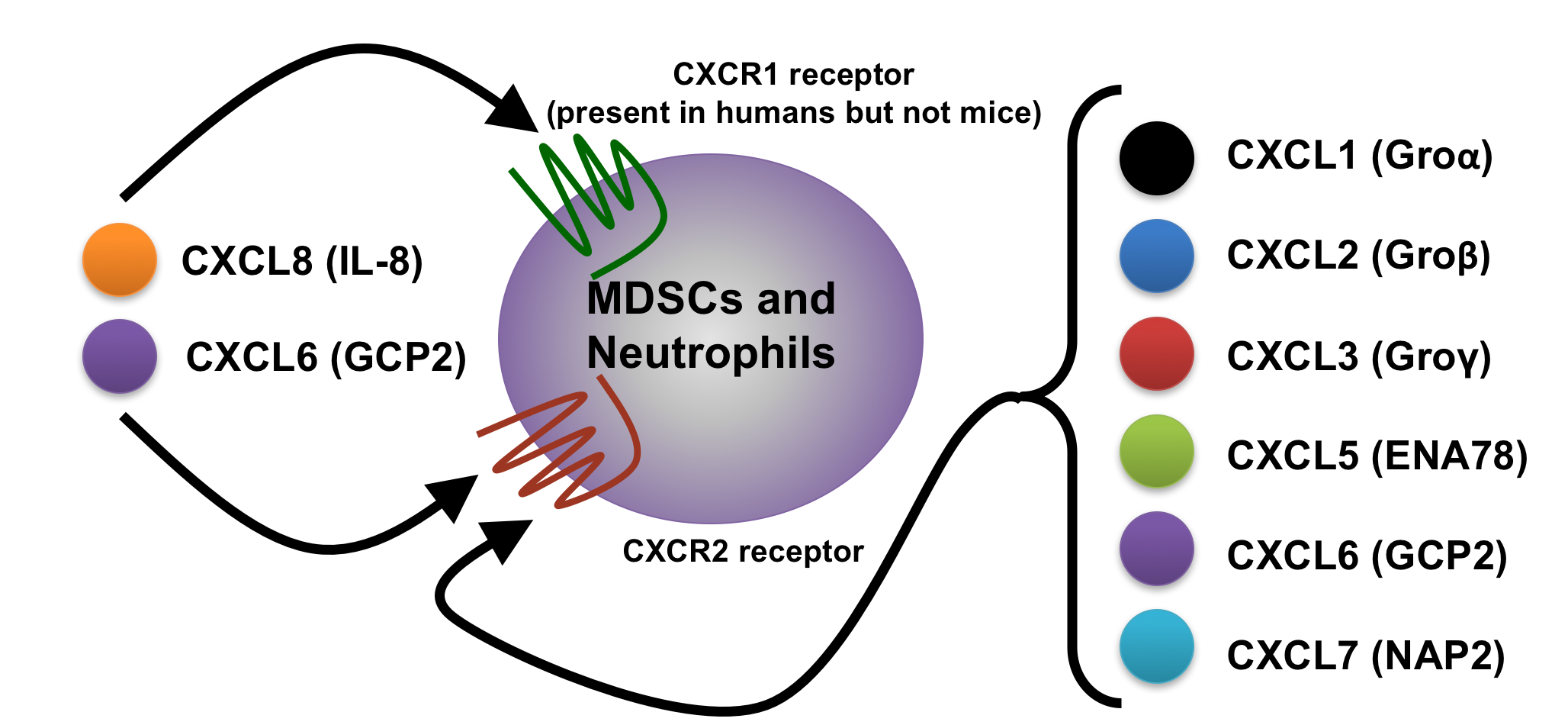
CXCR1 and CXCR2: Basic Receptor Biology
- CXCR1 and CXCR2 are G protein-coupled receptors that bind chemokines.
- CXCR1 and CXCR2 both bind CXCL6 (GCP2) and CXCL8 (IL-8) chemokines.
- CXCR2 also binds other chemokines (CXCL1, CXCL2, CXCL3, CXCL5 and CXCL7).
- Human tumors produce CXCR1 and CXCR2 chemokine ligands
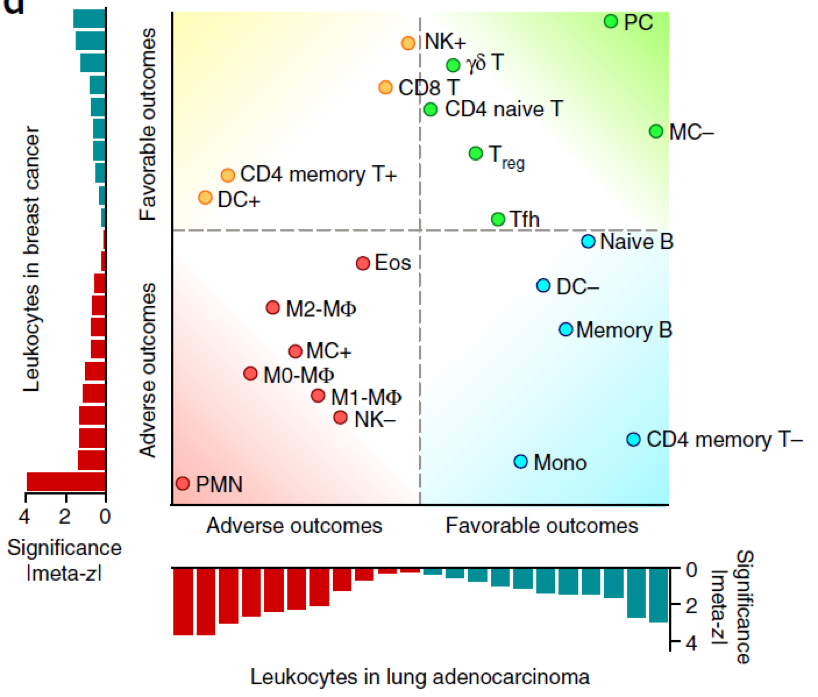
Neutrophils Are The Most Important Immuno-Suppressive Cell in Human Tumor Microenvironment:
- Expression signatures from ~18,000 human tumors with overall survival outcomes across 39 malignancies.
- Neutrophil (i.e., PMN) infiltration (arrow) stood alone as the factor providing the most adverseprognostic signal of any cell type in the tumor microenvironment (shown for breast cancer and lung adenocarcinoma).*
SX-682 Satisfies Lipinski Rules, Exhibits Nanomolar Potency, and is Highly Selective
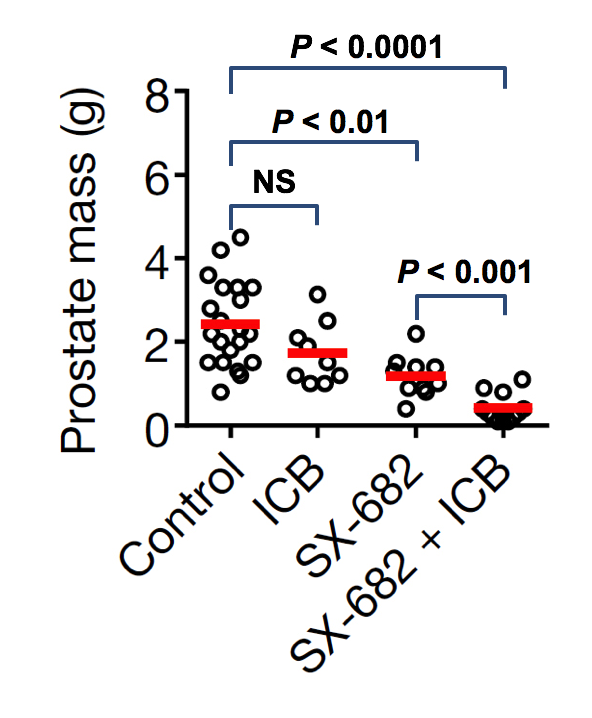
SX-682 Potently Synergized with Anti-PD1 and Anti-CTLA4 and Eliminated Lung Metastasis in mCRPC Model, Continued
SX-682 Clinical Development Plan Overview
- The Melanoma Phase I/II trial is funded under an SBIR grant from the NCI
- A CRADA recently awarded by the NCI will fund up to two additional Phase I and two Phase II trials in solid tumors; under the CRADA Syntrix will contribute SX-682 study drug and the NCI will be responsible for all other clinical trial expenses.
- $45M in Series A funding will accelerate the clinical trial plan and fund the extensive development program outlined above.
The health impact and toll of COPD.
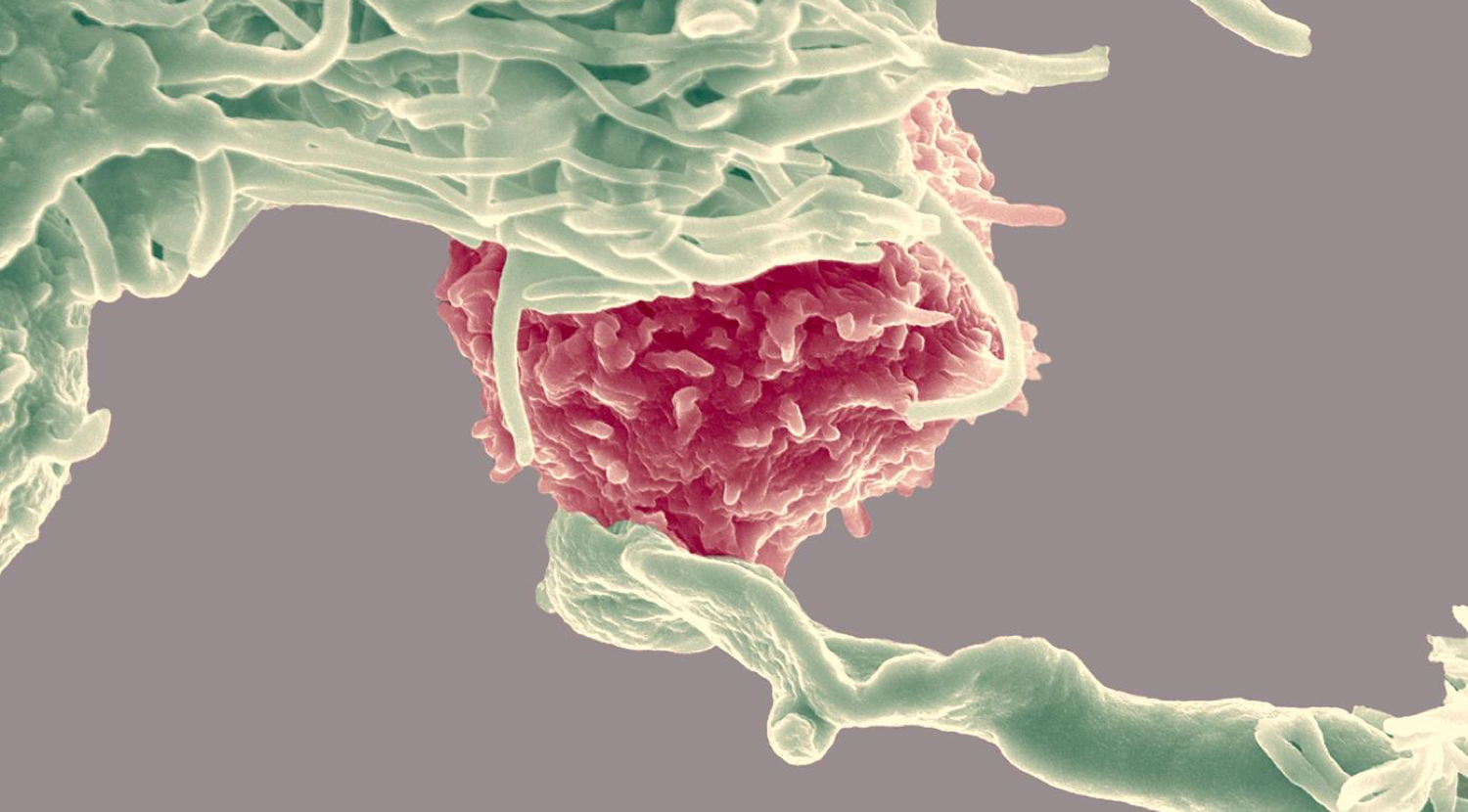
Neutrophils, a type of white blood cell or leukocyte, are key players in asthma pathology. This video shows how these cells reach their destination in the lung through a process of rolling, chemokine activation (e.g. CXCR1/2, a G protein coupled receptor), adhesion and translocation (together extravasation).
This is a text block. Click the edit button to change this text.
